Your Trusted Partner in End‑to‑End Pharmaceutical Development & Manufacturing
From formulation to packaging, Synokem delivers world‑class CDMO solutions that ensure speed, quality, and compliance — empowering pharma and biotech companies to bring life‑changing medicines to market faster.
INTERNATIONAL BUSINESSES
Expanding Horizons, Enabling Health Worldwide
Since it started its export operation in 2015 with a CDDA (Sri Lanka FDA authority) license, Synokem Pharmaceutical Limited has been developing its global reach in various continents in the past seven years. We have expanded into other markets by securing international regulatory approvals and passing various country-specific audits successfully. We continue to focus on conducting new stringent regulatory authority (SRA) audits with an objective to enter regulated markets.
REGULATORY & INNOVATION
With over 44 years of industry experience, Synokem combines scientific expertise, state-of-the-art R&D facilities, and heritage to deliver a vision-driven portfolio of off-patent molecules to the global healthcare sector.
GLOBAL PRESENCE
Serving markets across 6 continents with regulatory excellence
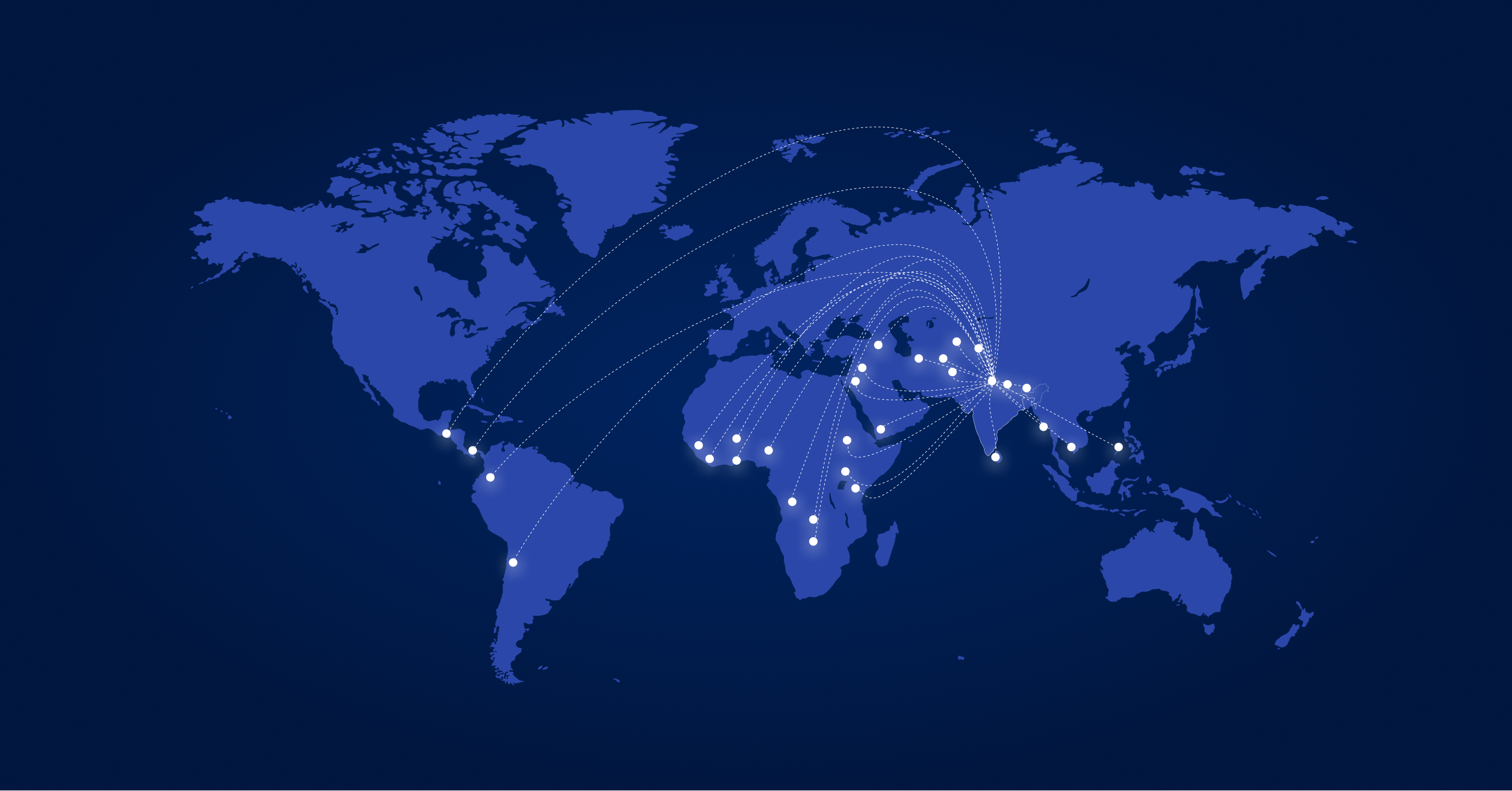
8 Countries
Kenya, Nigeria, Uganda, Sudan, Zambia, Botswana, DR Congo, Ghana
Upcoming: Tanzania, Ethiopia
4 Countries
Ecuador, Guatemala, Costa Rica, Venezuela
2 Countries
Yemen, Kurdistan
5 Countries
Uzbekistan, Kyrgyzstan, Tajikistan, Turkmenistan, Mongolia
4 Countries
Afghanistan, Bhutan, Nepal, Sri Lanka
3 Countries
Myanmar, Philippines, Cambodia
KEY APPROVALS
Regulatory excellence across global markets including:
OUR UNIQUE VALUE PROPOSITION
Regulatory Expertise
CTD dossiers, clinical trials, BE studies, WHO-GMP/ICH compliance, CQA audits, and quality agreements.
Customisation
Solutions customized to market requirements but keeping international standards.
Delivery
Timely delivery with strong commitment follow-through.
Quality
Adhering to specified standards of quality.
Innovation
Fully equipped R&D department, formulation development, microbiological & analytical testing, stability studies.
Recent Regulatory Developments (2025/26)
Country audit filings under preparation for Uzbekistan and Kyrgyzstan.
Application for EAEU audit by CIS central authority underway.
